Looking at how COVID-19 vaccine transportation and distribution create
health & safety challenges for air freight and logistics companies
According to the World Health Organization (WHO), as of June 2021, approximately 3 billion vaccine doses have been administered globally.1 This achievement is the result of a complicated supply chain network in which aviation plays a crucial role. DHL and McKinsey estimate that the delivery of 10 billion vaccine doses globally would require approximately 15,000 flights and 15 million cooling boxes.2
As mass vaccination against the coronavirus started, a key challenge has been to keep millions of doses of vaccines at the right temperature. An increase in temperature inside a truck or aircraft, by half a degree, for half an hour, would reportedly result in a 'defrosted' vaccine which has then to be discarded. One of the most widely used vaccines globally, Pfizer-BioNTech, requires a very low storage temperature of minus 70-80°C to remain stable and effective.3 Nonetheless, in May 2021, US, EU, and Canadian drug regulators allowed Pfizer to store the COVID-19 vaccine in a typical vaccine refrigerator for up to one month, thus minimizing the need to ship the vaccine in extremely low temperatures in dry ice.4,5 This highlights that better transportation technology and related, updated regulatory requirements for convenient storage can remove further logistical barriers to reaching a wider portion of the population.
One of the main factors affecting vaccine transportation is air transport safety
Before the pandemic, logistics companies were already transporting pharmaceutical products at extremely low temperatures in special containers with dry ice. The new challenges concerning refrigeration systems for transporting vaccines involved adapting to existing cooling technologies as quickly as possible with specific provisions. Requirements for consideration included package size, storage, and transport times, while ensuring high safety standards for a growing number of logistic workers, drivers, pilots and crews newly engaged in vaccine transportation.
Dry ice presents several safety challenges. It is the frozen form of carbon dioxide (CO2) and is unsafe when it is sublimated, i.e., transformed into a vapor state, bypassing the liquid state. When this occurs, carbon dioxide is produced, and the air becomes potentially toxic for pilots and crews. In poorly ventilated rooms, a concentration of above 10% can lead to asphyxiation.6 Another challenge is that the sublimation process can create pressure in the container, leading to a risk of explosion.
Vaccine transport staff should be familiar with dry ice handling guidelines
The European Aviation Safety Agency (EASA) has issued guidance on the use of dry ice for storing COVID-19 vaccines.6 The recommendations came together with the US Federal Aviation Administration’s (FAA) Safety Alert for Operators (SAFO), outlining safety precautions that ground and flight crews should take when preparing to transport the vaccine.7 Boeing, Airbus, WHO as well as local authorities of different countries also issued guidelines.8
There is limited visibility on occupational health and safety (H&S) incidents related to handling dry ice as of the vaccine transportation process. This is because most companies report on overall quantitative figures on their occupational accidents and/or injury rates. In general, air freight and logistics companies, especially logistics giants (the “big three”) FedEx, UPS, and DHL, tend to effectively manage their occupational health and safety risks, with 86% of the companies in the overall sector showing negligible to low risk scores under Sustainalytics’ ESG Risk Ratings for the Occupational Health and Safety Material ESG Issue, as illustrated in Figure 1.
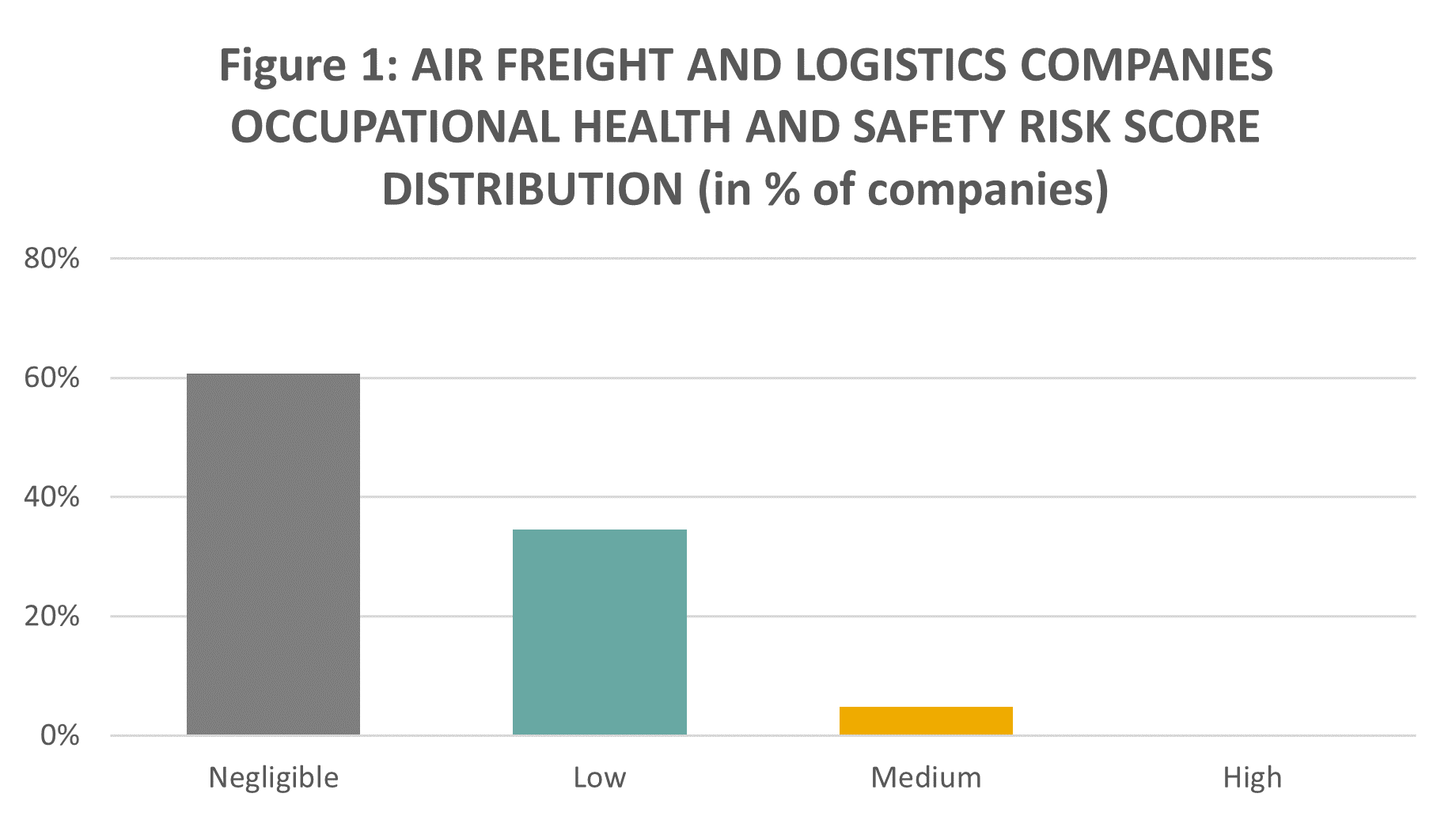
Source: Sustainalytics
Commercial passenger airlines have also entered the race to transport vaccines. For example, United, Delta, Lufthansa, IAG and Air France-KLM joined forces to deliver vaccines.9 Such involvement benefits the airlines by allowing otherwise grounded planes and crews to generate income and minimise to a degree the impact of the pandemic on battered revenues, (airline financial losses globally resulting from COVID-19 impacts reached USD 370 billion in 2020).10 Another potential benefit for an airline assisting with vaccine distribution is that increased vaccination will lead to the faster reopening of borders and reduced travel restrictions, leading to a speedier recovery for the airline industry.11 And to meet the challenge of transporting the millions of doses of vaccines needed around the globe, airlines have been converting some passenger dedicated space on airplanes to vaccine transportation, which constitutes further logistical hurdles.
Overall, compared with air freight and logistics companies, we note lower management scores by airlines, with more than 60% of companies displaying average management of the issue. This may point to a lesser preparedness of these companies to tackle further, newer H&S risks related to vaccine transportation, which for most airlines, falls to a great extent outside of their classical cargo transportation capabilities.
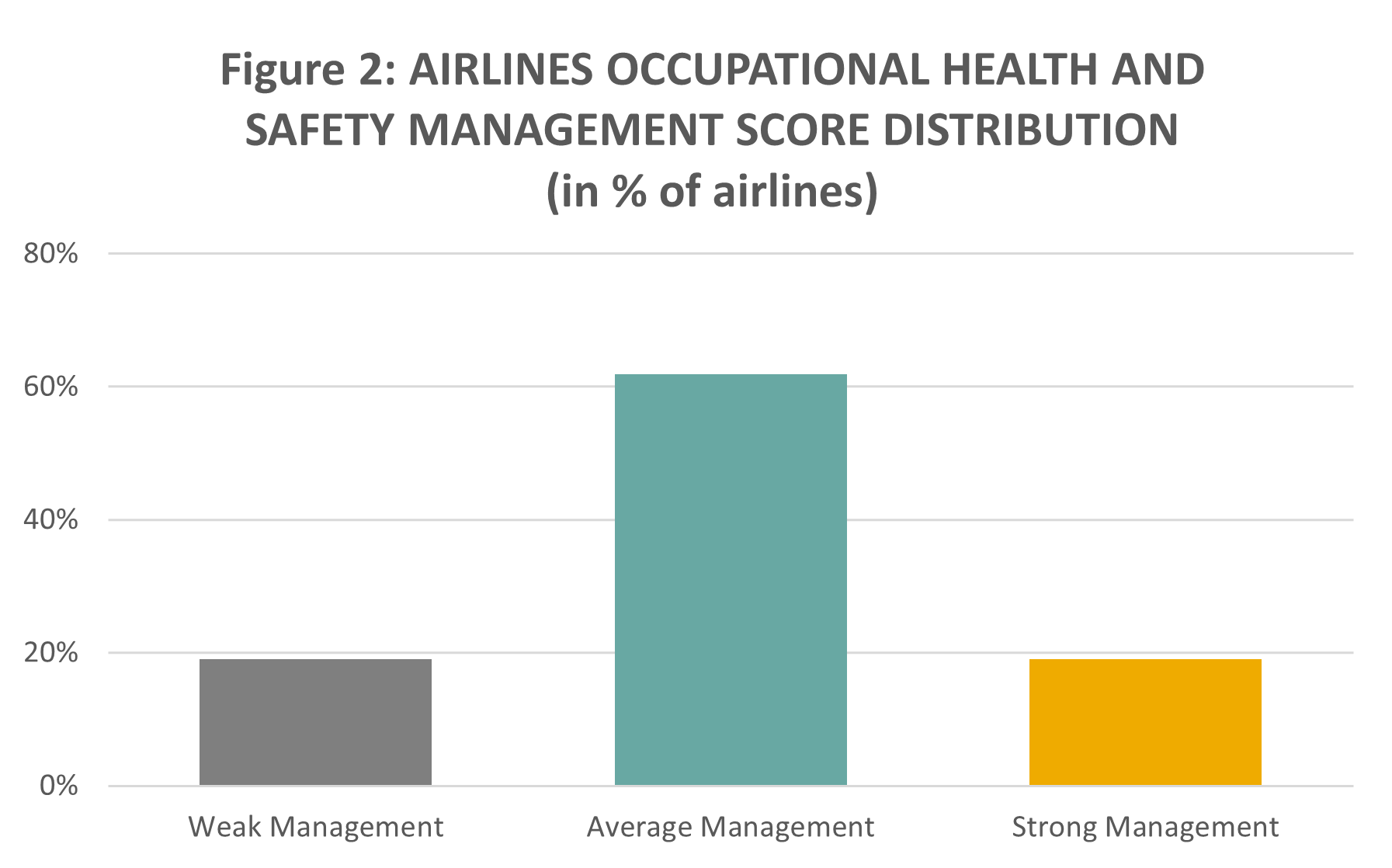
Source: Sustainalytics
While industry leaders generally show good performance when it comes to H&S issues, the risk might be more substantial for smaller, regional carriers who still must use dry ice for vaccines to reach remote areas with poor transport and storage infrastructures. Handling hazardous materials, such as dry ice, requires robust dedicated health and safety programmes, including special training for employees, risk assessments and emergency preparedness procedures, especially for those regional airlines more specialized in carrying passengers and less in air cargo transportation.
Learn more about how Sustainalytics’ ESG Risk Ratings can support investors with complex considerations in their portfolio selection.
.png?Status=Master&sfvrsn=f46cedf4_1)
Sources:
- https://covid19.who.int/
- https://www.dpdhl.com/en/media-relations/press-releases/2020/delivery-covid-19-vaccine-dhl-study-shows-how-public-and-private-sector-can-partner-for-success.html
- https://www.pfizer.com/news/hot-topics/covid_19_vaccine_u_s_distribution_fact_sheet
- https://www.reuters.com/business/healthcare-pharmaceuticals/pfizer-covid-19-vaccine-can-be-stored-refrigerator-month-us-says-2021-05-20/
- https://www.reuters.com/business/healthcare-pharmaceuticals/eu-regulator-backs-longer-storage-time-pfizer-vaccine-normal-fridges-2021-05-17/
- https://www.easa.europa.eu/newsroom-and-events/press-releases/easa-issues-guidance-use-dry-ice-covid-19-vaccine-transportation
- https://www.faa.gov/other_visit/aviation_industry/airline_operators/airline_safety/safo/all_safos/
- https://www.who.int/publications/i/item/who-2019-ncov-vaccine-deployment-logistics-2021-1
- https://www.flightglobal.com/flight-international/airline-industry-embarks-on-global-vaccine-challenge/142007.article
- https://www.icao.int/Newsroom/Pages/2020-passenger-totals-drop-60-percent-as-COVID19-assault-on-international-mobility-continues.aspx
- https://www.iata.org/en/pressroom/pr/2021-05-19-01/
Recent Content
SFDR 2.0: Navigating the Shift from Complexity to Clarity in Sustainable Investing
This article highlights the potential implications changes to SFDR 2.0 could create for asset managers and institutional investors as well as the growing role of high-quality, independent data in navigating this regulatory transition.



.tmb-thumbnl_rc.png?Culture=en&sfvrsn=9af6b902_1)
